Day 1 Due 11/14 (A) 11/15 (B)AT HOME
|
3.1 - Weather Patterns |
Introduction
Wind, precipitation, warming, and cooling depend on how much energy is in the atmosphere and where that energy is located. Much more energy from the Sun reaches low latitudes (nearer the equator) than high latitudes (nearer the poles). These differences in insolation –- the amount of solar radiation that reaches a given area in a given time -- cause the winds, affect climate, and drive ocean currents. Heat is held in the atmosphere by greenhouse gases.
Energy, Temperature, and Heat
Energy
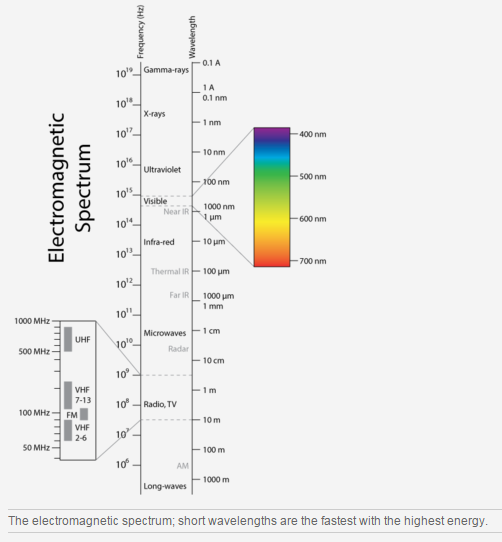
Energy travels through space or material. This is obvious when you stand near a fire and feel its warmth or when you pick up the handle of a metal pot even though the handle is not sitting directly on the hot stove. Invisible energy waves can travel through air, glass, and even the vacuum of outer space. These waves have electrical and magnetic properties, so they are called electromagnetic waves. The transfer of energy from one object to another through electromagnetic waves is known as radiation.
Different wavelengths of energy create different types of electromagnetic waves (Figure below).
Wind, precipitation, warming, and cooling depend on how much energy is in the atmosphere and where that energy is located. Much more energy from the Sun reaches low latitudes (nearer the equator) than high latitudes (nearer the poles). These differences in insolation –- the amount of solar radiation that reaches a given area in a given time -- cause the winds, affect climate, and drive ocean currents. Heat is held in the atmosphere by greenhouse gases.
Energy, Temperature, and Heat
Energy
Energy travels through space or material. This is obvious when you stand near a fire and feel its warmth or when you pick up the handle of a metal pot even though the handle is not sitting directly on the hot stove. Invisible energy waves can travel through air, glass, and even the vacuum of outer space. These waves have electrical and magnetic properties, so they are called electromagnetic waves. The transfer of energy from one object to another through electromagnetic waves is known as radiation.
Different wavelengths of energy create different types of electromagnetic waves (Figure below).
- The wavelengths humans can see are known as "visible light." These wavelengths appear to us as the colors of the rainbow. What objects can you think of that radiate visible light? Two include the Sun and a light bulb.
- The longest wavelengths of visible light appear red. Infrared wavelengths are longer than visible red. Snakes can see infrared energy. We feel infrared energy as heat.
- Wavelengths that are shorter than violet are called ultraviolet.
Can you think of some objects that appear to radiate visible light, but actually do not? The moon and the planets do not emit light of their own; they reflect the light of the Sun. Reflection is when light (or another wave) bounces back from a surface.Albedo is a measure of how well a surface reflects light. A surface with high albedo reflects a large percentage of light. A snow field has high albedo.
One important fact to remember is that energy cannot be created or destroyed -- it can only be changed from one form to another. This is such a fundamental fact of nature that it is a law: the law of conservation of energy.
In photosynthesis, for example, plants convert solar energy into chemical energy that they can use. They do not create new energy. When energy is transformed, some nearly always becomes heat. Heat transfers between materials easily, from warmer objects to cooler ones. If no more heat is added, eventually all of a material will reach the same temperature.
Temperature
Temperature is a measure of how fast the atoms in a material are vibrating. High temperature particles vibrate faster than low temperature particles. Rapidly vibrating atoms smash together, which generates heat. As a material cools down, the atoms vibrate more slowly and collide less frequently. As a result, they emit less heat. What is the difference between heat and temperature?
Heat
Heat is taken in or released when an object changes state, or changes from a gas to a liquid, or a liquid to a solid. This heat is called latent heat. When a substance changes state, latent heat is released or absorbed. A substance that is changing its state of matter does not change temperature. All of the energy that is released or absorbed goes toward changing the material’s state.
For example, imagine a pot of boiling water on a stove burner: that water is at 100°C (212°F). If you increase the temperature of the burner, more heat enters the water. The water remains at its boiling temperature, but the additional energy goes into changing the water from liquid to gas. With more heat the water evaporates more rapidly. When water changes from a liquid to a gas it takes in heat. Since evaporation takes in heat, this is called evaporative cooling. Evaporative cooling is an inexpensive way to cool homes in hot, dry areas.
Substances also differ in their specific heat, the amount of energy needed to raise the temperature of one gram of the material by 1.0°C (1.8°F). Water has a very high specific heat, which means it takes a lot of energy to change the temperature of water. Let's compare a puddle and asphalt, for example. If you are walking barefoot on a sunny day, which would you rather walk across, the shallow puddle or an asphalt parking lot? Because of its high specific heat, the water stays cooler than the asphalt, even though it receives the same amount of solar radiation.
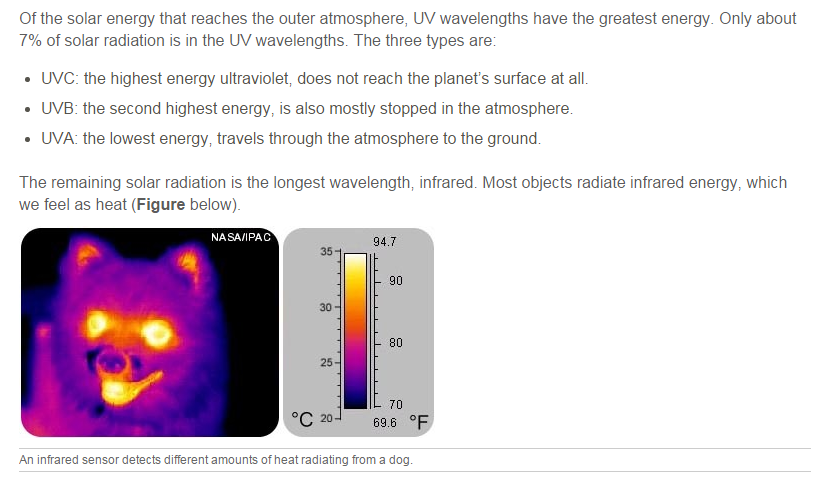
Energy From the SunMost of the energy that reaches the Earth’s surface comes from the Sun (Figure below). About 44% of solar radiation is in the visible light wavelengths, but the Sun also emits infrared, ultraviolet, and other wavelengths.
One important fact to remember is that energy cannot be created or destroyed -- it can only be changed from one form to another. This is such a fundamental fact of nature that it is a law: the law of conservation of energy.
In photosynthesis, for example, plants convert solar energy into chemical energy that they can use. They do not create new energy. When energy is transformed, some nearly always becomes heat. Heat transfers between materials easily, from warmer objects to cooler ones. If no more heat is added, eventually all of a material will reach the same temperature.
Temperature
Temperature is a measure of how fast the atoms in a material are vibrating. High temperature particles vibrate faster than low temperature particles. Rapidly vibrating atoms smash together, which generates heat. As a material cools down, the atoms vibrate more slowly and collide less frequently. As a result, they emit less heat. What is the difference between heat and temperature?
- Temperature measures how fast a material’s atoms are vibrating.
- Heat measures the material’s total energy.
- The flame has higher temperature, but less heat, because the hot region is very small.
- The bathtub has lower temperature but contains much more heat because it has many more vibrating atoms. The bathtub has greater total energy.
Heat
Heat is taken in or released when an object changes state, or changes from a gas to a liquid, or a liquid to a solid. This heat is called latent heat. When a substance changes state, latent heat is released or absorbed. A substance that is changing its state of matter does not change temperature. All of the energy that is released or absorbed goes toward changing the material’s state.
For example, imagine a pot of boiling water on a stove burner: that water is at 100°C (212°F). If you increase the temperature of the burner, more heat enters the water. The water remains at its boiling temperature, but the additional energy goes into changing the water from liquid to gas. With more heat the water evaporates more rapidly. When water changes from a liquid to a gas it takes in heat. Since evaporation takes in heat, this is called evaporative cooling. Evaporative cooling is an inexpensive way to cool homes in hot, dry areas.
Substances also differ in their specific heat, the amount of energy needed to raise the temperature of one gram of the material by 1.0°C (1.8°F). Water has a very high specific heat, which means it takes a lot of energy to change the temperature of water. Let's compare a puddle and asphalt, for example. If you are walking barefoot on a sunny day, which would you rather walk across, the shallow puddle or an asphalt parking lot? Because of its high specific heat, the water stays cooler than the asphalt, even though it receives the same amount of solar radiation.
Energy From the SunMost of the energy that reaches the Earth’s surface comes from the Sun (Figure below). About 44% of solar radiation is in the visible light wavelengths, but the Sun also emits infrared, ultraviolet, and other wavelengths.
Solar Radiation on Earth
Different parts of the Earth receive different amounts of solar radiation. Which part of the planet receives the most insolation? The Sun's rays strike the surface most directly at the equator.
Different areas also receive different amounts of sunlight in different seasons. What causes the seasons? The seasons are caused by the direction Earth's axis is pointing relative to the Sun.
The Earth revolves around the Sun once each year and spins on its axis of rotation once each day. This axis of rotation is tilted 23.5° relative to its plane of orbit around the Sun. The axis of rotation is pointed toward Polaris, the North Star. As the Earth orbits the Sun, the tilt of Earth's axis stays lined up with the North Star.
Different parts of the Earth receive different amounts of solar radiation. Which part of the planet receives the most insolation? The Sun's rays strike the surface most directly at the equator.
Different areas also receive different amounts of sunlight in different seasons. What causes the seasons? The seasons are caused by the direction Earth's axis is pointing relative to the Sun.
The Earth revolves around the Sun once each year and spins on its axis of rotation once each day. This axis of rotation is tilted 23.5° relative to its plane of orbit around the Sun. The axis of rotation is pointed toward Polaris, the North Star. As the Earth orbits the Sun, the tilt of Earth's axis stays lined up with the North Star.
Heat at Earth’s Surface
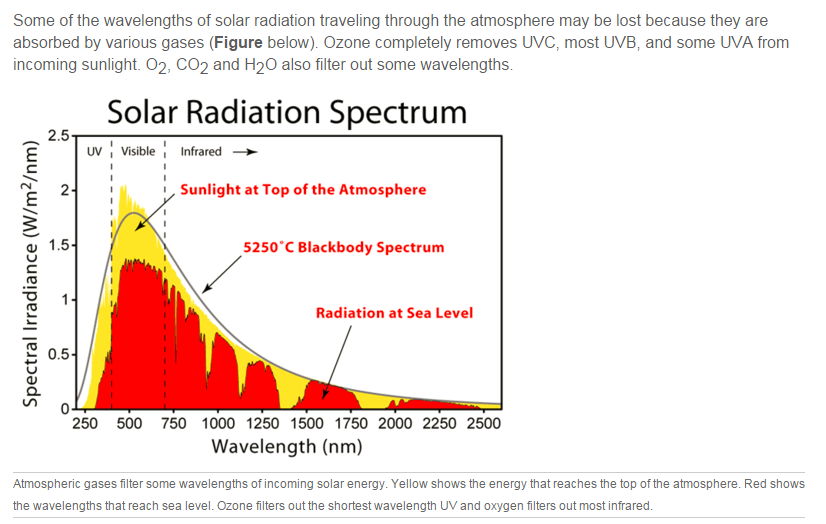
About half of the solar radiation that strikes the top of the atmosphere is filtered out before it reaches the ground. This energy can be absorbed by atmospheric gases, reflected by clouds, or scattered. Scattering occurs when a light wave strikes a particle and bounces off in some other direction.
About 3% of the energy that strikes the ground is reflected back into the atmosphere. The rest is absorbed by rocks, soil, and water and then radiated back into the air as heat. These infrared wavelengths can only be seen by infrared sensors.
Because solar energy continually enters Earth’s atmosphere and ground surface, is the planet getting hotter? The answer is no (although there is an exception) because energy from Earth escapes into space through the top of the atmosphere. If the amount that exits is equal to the amount that comes in, then average global temperature stays the same. This means that the planet’s heat budget is in balance. What happens if more energy comes in than goes out? If more energy goes out than comes in?
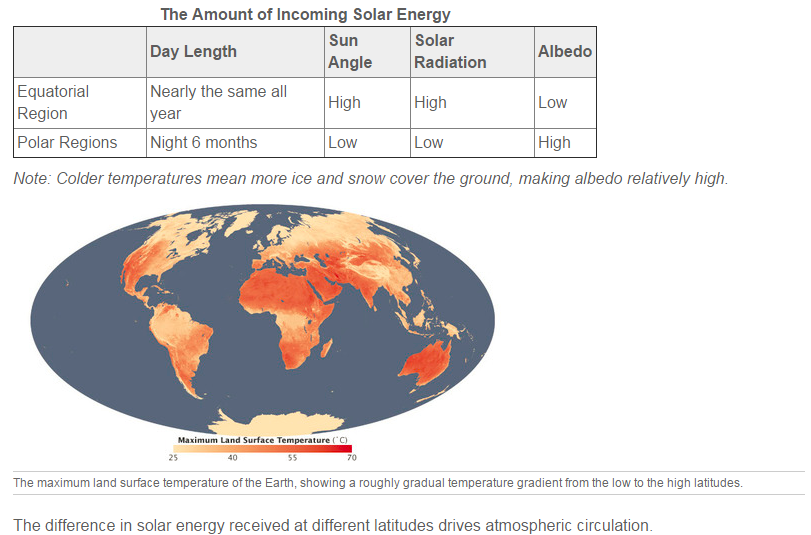
To say that the Earth’s heat budget is balanced ignores an important point. The amount of incoming solar energy is different at different latitudes (Figure below). Where do you think the most solar energy ends up and why? Where does the least solar energy end up and why? See Table below
About half of the solar radiation that strikes the top of the atmosphere is filtered out before it reaches the ground. This energy can be absorbed by atmospheric gases, reflected by clouds, or scattered. Scattering occurs when a light wave strikes a particle and bounces off in some other direction.
About 3% of the energy that strikes the ground is reflected back into the atmosphere. The rest is absorbed by rocks, soil, and water and then radiated back into the air as heat. These infrared wavelengths can only be seen by infrared sensors.
Because solar energy continually enters Earth’s atmosphere and ground surface, is the planet getting hotter? The answer is no (although there is an exception) because energy from Earth escapes into space through the top of the atmosphere. If the amount that exits is equal to the amount that comes in, then average global temperature stays the same. This means that the planet’s heat budget is in balance. What happens if more energy comes in than goes out? If more energy goes out than comes in?
To say that the Earth’s heat budget is balanced ignores an important point. The amount of incoming solar energy is different at different latitudes (Figure below). Where do you think the most solar energy ends up and why? Where does the least solar energy end up and why? See Table below
The Greenhouse Effect
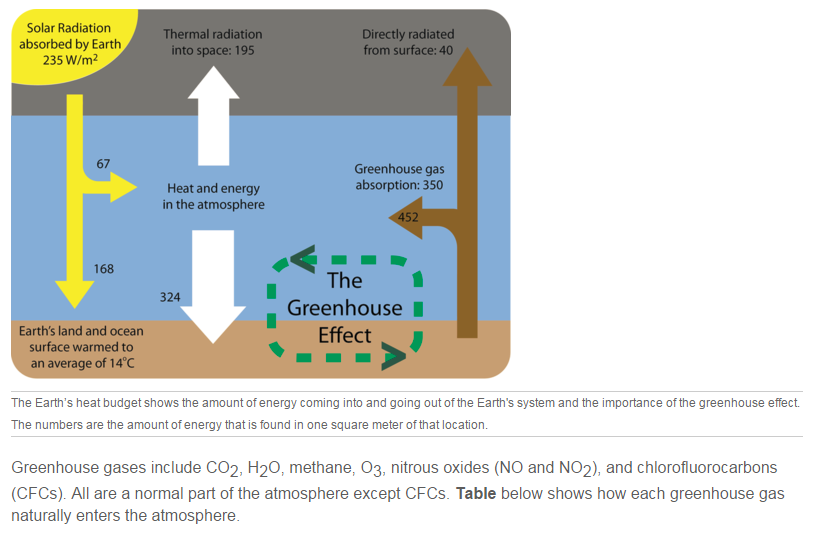
The exception to Earth’s temperature being in balance is caused by greenhouse gases. But first the role of greenhouse gases in the atmosphere must be explained. Greenhouse gases warm the atmosphere by trapping heat. Some of the heat radiation out from the ground is trapped by greenhouse gases in the troposphere. Like a blanket on a sleeping person, greenhouse gases act as insulation for the planet. The warming of the atmosphere because of insulation by greenhouse gases is called the greenhouse effect (Figure below). Greenhouse gases are the component of the atmosphere that moderate Earth’s temperatures.
The exception to Earth’s temperature being in balance is caused by greenhouse gases. But first the role of greenhouse gases in the atmosphere must be explained. Greenhouse gases warm the atmosphere by trapping heat. Some of the heat radiation out from the ground is trapped by greenhouse gases in the troposphere. Like a blanket on a sleeping person, greenhouse gases act as insulation for the planet. The warming of the atmosphere because of insulation by greenhouse gases is called the greenhouse effect (Figure below). Greenhouse gases are the component of the atmosphere that moderate Earth’s temperatures.
Questions
1. Why do the polar regions have high albedo?
2. On a map of average annual temperature, why are the lower latitudes so much warmer than the higher latitudes?
3. Why is carbon dioxide the most important greenhouse gas?
4. How does the amount of greenhouse gases in the atmosphere affect the atmosphere’s temperature?
5. How does the difference in solar radiation that reaches the lower and upper latitudes explain the way the atmosphere circulates?
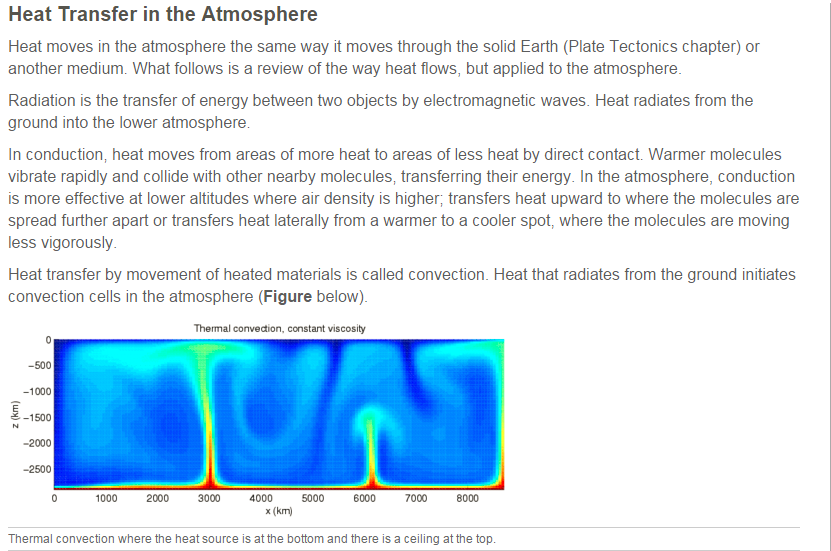
6. Describe the three ways heat is transferred through the atmosphere.
1. Why do the polar regions have high albedo?
2. On a map of average annual temperature, why are the lower latitudes so much warmer than the higher latitudes?
3. Why is carbon dioxide the most important greenhouse gas?
4. How does the amount of greenhouse gases in the atmosphere affect the atmosphere’s temperature?
5. How does the difference in solar radiation that reaches the lower and upper latitudes explain the way the atmosphere circulates?
6. Describe the three ways heat is transferred through the atmosphere.
IN CLASS